Neumous Helps Treat Brain Diseases with focused ultrasound that open up the Blood-Brain Barrier.
Neumous Helps Treat Brain Diseases with focused ultrasound that open up the Blood-Brain Barrier.
Posted July. 10, 2025 10:29,
Updated July. 10, 2025 10:35
- Neumous, has developed a technology that uses focused ultrasound to
temporarily open the BBB, allowing drugs to pass through and treat brain
diseases such as Alzheimer's and Parkinson's.
- Neumous’s non-invasive technology is cost-effective and safe, avoiding the
complexities of surgery and enabling precise control over the BBB opening and
closing.
- Neumous has gained recognition both domestically and internationally,
securing funding and technological support to advance its innovative approach
to treating neurological disorders.
Brain diseases such as brain tumors, Alzheimer's disease, Parkinson's disease, and ALS(Amyotrophic Lateral Sclerosis) are known to be extremely difficult to treat. This is due to the Blood-Brain Barrier(BBB), which is located between the brain and blood vessels and blocks the entry of drugs and other therapeutic agents To this day, numerous treatments for brain diseases have been developed, but most have failed to penetrate the BBB and, as a result, have not proven effective.
While the pharmaceutical industry continues to research and develop treatments for brain diseases that operate in various ways, other approach approaches are being explored as well. One such approach involves technologies that induce the BBB to accept drugs and facilitate their delivery throughout the cerebrovascular system. Neumous, a Korean medical device startup, has made a notable impact in this field.

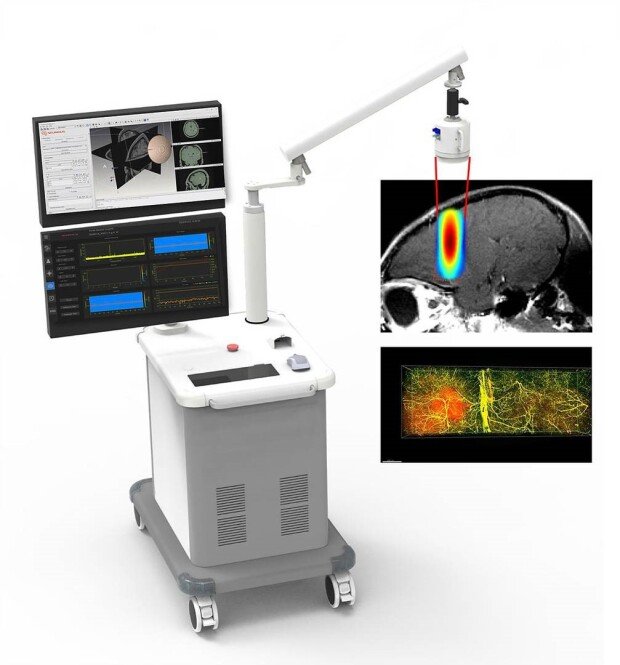
Neumous has developed a technology to control the BBB through focused ultrasound. The technology works by concentrating ultrasound waves and delivering them to a specific area of the brain, where they apply mechanical stimulation to the blood vessels. This stimulation loosens the structure of certain proteins that make up the BBB. The core principle of Neumous' technology is to enable drugs to pass through the temporarily opened gaps.
Building on its core technology for controlling the BBB with focused ultrasound, Neumous has developed an even more advanced capability: opening and closing the BBB in targeted brain areas for a precisely controlled duration. This enables the treatment to be tailored to the location and characteristics of the brain lesion, as well as the type and dosage of the drug, thereby maximizing therapeutic efficacy. As a non-invasive technology, the procedure is straightforward, avoiding the complexities of surgery. Its versatility also makes the technology applicable to a wide range of neurological disorders."

Despite the successful completion of safety, Phase I, and Phase II trials, numerous therapeutic candidates for brain diseases have been halted due to their failure to penetrate the BBB. Neumous’ technology enables these neurotherapeutics to achieve their intended efficacy. Its function extends beyond delivering conventional drugs to transporting gene therapies, cell therapies, and even contrast agents into the brain to support the diagnosis and treatment of neurological disorders.
Ju-young Park, CEO of Neumous, has long been engaged in research on drug delivery to organs such as the brain's blood vessels and the retina. He began R&D on focused ultrasound technology, which targets the BBB, in earlier years. He became determined to bring his research into reality after witnessing the suffering of brain tumor patients, over 90% of whom experience recurrence—and the tragic deaths of young pediatric patients with brain diseases.
After founding Neumous, Dr. Park began assembling his team, bringing on board ultrasound hardware and software developers, medical device designers, and specialists in regulatory affairs. Soon after, researchers in neurology and seasoned professionals from the pharmaceutical sector joined Neumous, lending their expertise to the mission. Each member is an expert with over 15 years of experience in their respective fields, united by a shared vision: to help patients with brain diseases and bring a new ‘light’ to the industry. This mission is even reflected in the company's name. ‘Neumous’ is inspired by 'Lumos,' the light-creating magic spell from the popular ‘Harry Potter’series.
Companies and organizations share Neumous’ values have steadily come forward to collaborate. The Chungbuk Center for Creative Economy and Innovation (CCEI) is a prime example. Although Neumous is a medical device startup, where collaboration with the pharmaceutical and biotechnology sectors is essential due to the nature of its business. Chungbuk CCEI has played a key role in building the necessary networks and partnerships. It has also supported the company by developing promotional strategies, helping to establish patents, and facilitating collaborations with global pharmaceutical companies efforts aimed at turning Neumos’ technology into reality and bringing it to both domestic and international markets.

Due to these efforts, Neumous has successfully advanced its focused ultrasound technology and achieved clear differentiation from competing BBB opening technologies. 'Brain Tissue Thermal Surgery' and 'Brain Implants' are techniques that physically open the BBB. The former has been approved by the U.S. FDA for the treatment of Parkinson's disease. However, it requires a dedicated MRI costing several billion won and the procedure takes several hours. Brain implants involve inserting devices into the skulls of brain tumor patients. This technique can only be applied to deep brain regions and carries concerns about safety.
Neumous' technology can be implemented at one-tenth the cost of existing methods. It is considered safe, as it does not involve surgery. Park emphasizes that no tissue damage or inflammation was observed when the technology was tested in the brain of a monkey, a primate closely related to humans. The procedure is also brief, taking approximately 15 minutes.
Furthermore, Neumous has refined its technology to support nearly every stage of drug development. Whether in preclinical trials, animal testing, or clinical trials, the technology is utilized to open the BBB and deliver drugs to the brain. This versatility allows Neumous to effectively respond to the various needs of pharmaceutical companies.
In just three years since its founding, Neumous has gained recognition both domestically and internationally. The company secured a large-scale national project obtaining both funding and technological support, and won the top prize at the K-Startup “King of Kings” Challenge. It was also recognized as an innovative medical device. The Korea Innovative Medicines Consortium (KIMCo) recognizing its achievements, selected Neumous as a company to nurture. Recently, Neumous successfully completed its Series -A funding round. Building on these achievements and promising experimental results, the company now plans to complete clinical trials in a short timeframe and expand onto the global stage.

Neumous is set to complete its clinical trials and release the results globally within this year. Following pre-clinical animal studies, the company confirmed that its technology can open the BBB safely and consistently. Once the clinical results become available, Neumous plans to promptly share its findings with the global pharmaceutical industry and begin the production and supply of devices tailored to client needs.
Dr. Park stated, "Neumous, which began as a research-based initiative, has now grown into a medical device startup with the capability to serve as a partner to pharmaceutical companies. True to our name, we aim to become a company that brings new ‘light’ to patients with brain diseases and advances in brain treatment. We deeply empathize with the suffering of patients and their families, and we are committed to offering them new hope and effective solutions."
By Ju Kyung Cha (racingcar@itdonga.com)
temporarily open the BBB, allowing drugs to pass through and treat brain
diseases such as Alzheimer's and Parkinson's.
- Neumous’s non-invasive technology is cost-effective and safe, avoiding the
complexities of surgery and enabling precise control over the BBB opening and
closing.
- Neumous has gained recognition both domestically and internationally,
securing funding and technological support to advance its innovative approach
to treating neurological disorders.
Brain diseases such as brain tumors, Alzheimer's disease, Parkinson's disease, and ALS(Amyotrophic Lateral Sclerosis) are known to be extremely difficult to treat. This is due to the Blood-Brain Barrier(BBB), which is located between the brain and blood vessels and blocks the entry of drugs and other therapeutic agents To this day, numerous treatments for brain diseases have been developed, but most have failed to penetrate the BBB and, as a result, have not proven effective.
While the pharmaceutical industry continues to research and develop treatments for brain diseases that operate in various ways, other approach approaches are being explored as well. One such approach involves technologies that induce the BBB to accept drugs and facilitate their delivery throughout the cerebrovascular system. Neumous, a Korean medical device startup, has made a notable impact in this field.

Ju Young Park, CEO of Neumous introduces his company at the 2024 K-Startup Challenge / source= Neumous
Neumous has developed a technology to control the BBB through focused ultrasound. The technology works by concentrating ultrasound waves and delivering them to a specific area of the brain, where they apply mechanical stimulation to the blood vessels. This stimulation loosens the structure of certain proteins that make up the BBB. The core principle of Neumous' technology is to enable drugs to pass through the temporarily opened gaps.
Building on its core technology for controlling the BBB with focused ultrasound, Neumous has developed an even more advanced capability: opening and closing the BBB in targeted brain areas for a precisely controlled duration. This enables the treatment to be tailored to the location and characteristics of the brain lesion, as well as the type and dosage of the drug, thereby maximizing therapeutic efficacy. As a non-invasive technology, the procedure is straightforward, avoiding the complexities of surgery. Its versatility also makes the technology applicable to a wide range of neurological disorders."

BBB in the brain can be controlled in real time with focused ultrasound / source= Neumous
Despite the successful completion of safety, Phase I, and Phase II trials, numerous therapeutic candidates for brain diseases have been halted due to their failure to penetrate the BBB. Neumous’ technology enables these neurotherapeutics to achieve their intended efficacy. Its function extends beyond delivering conventional drugs to transporting gene therapies, cell therapies, and even contrast agents into the brain to support the diagnosis and treatment of neurological disorders.
Ju-young Park, CEO of Neumous, has long been engaged in research on drug delivery to organs such as the brain's blood vessels and the retina. He began R&D on focused ultrasound technology, which targets the BBB, in earlier years. He became determined to bring his research into reality after witnessing the suffering of brain tumor patients, over 90% of whom experience recurrence—and the tragic deaths of young pediatric patients with brain diseases.

Neumous's focused ultrasound instrument / source= Neumous
After founding Neumous, Dr. Park began assembling his team, bringing on board ultrasound hardware and software developers, medical device designers, and specialists in regulatory affairs. Soon after, researchers in neurology and seasoned professionals from the pharmaceutical sector joined Neumous, lending their expertise to the mission. Each member is an expert with over 15 years of experience in their respective fields, united by a shared vision: to help patients with brain diseases and bring a new ‘light’ to the industry. This mission is even reflected in the company's name. ‘Neumous’ is inspired by 'Lumos,' the light-creating magic spell from the popular ‘Harry Potter’series.
Companies and organizations share Neumous’ values have steadily come forward to collaborate. The Chungbuk Center for Creative Economy and Innovation (CCEI) is a prime example. Although Neumous is a medical device startup, where collaboration with the pharmaceutical and biotechnology sectors is essential due to the nature of its business. Chungbuk CCEI has played a key role in building the necessary networks and partnerships. It has also supported the company by developing promotional strategies, helping to establish patents, and facilitating collaborations with global pharmaceutical companies efforts aimed at turning Neumos’ technology into reality and bringing it to both domestic and international markets.

Dr. Park introduces Neumous at the 2024 Startup-Bus Joint IR / source=Neumous
Due to these efforts, Neumous has successfully advanced its focused ultrasound technology and achieved clear differentiation from competing BBB opening technologies. 'Brain Tissue Thermal Surgery' and 'Brain Implants' are techniques that physically open the BBB. The former has been approved by the U.S. FDA for the treatment of Parkinson's disease. However, it requires a dedicated MRI costing several billion won and the procedure takes several hours. Brain implants involve inserting devices into the skulls of brain tumor patients. This technique can only be applied to deep brain regions and carries concerns about safety.
Neumous' technology can be implemented at one-tenth the cost of existing methods. It is considered safe, as it does not involve surgery. Park emphasizes that no tissue damage or inflammation was observed when the technology was tested in the brain of a monkey, a primate closely related to humans. The procedure is also brief, taking approximately 15 minutes.

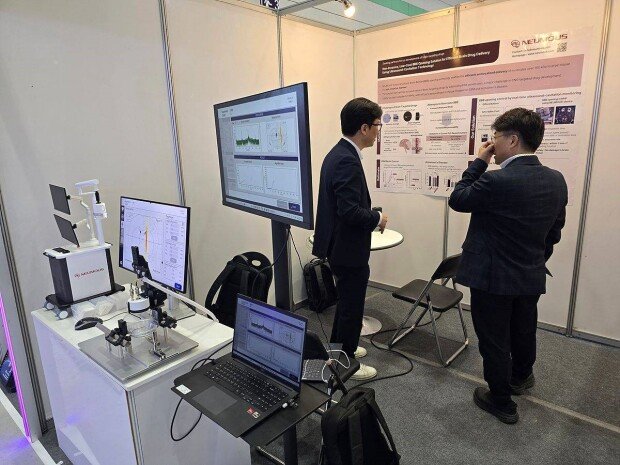
Neumous exhibites its technology and devices at BIO KOREA 2025 / source=Neumous
Furthermore, Neumous has refined its technology to support nearly every stage of drug development. Whether in preclinical trials, animal testing, or clinical trials, the technology is utilized to open the BBB and deliver drugs to the brain. This versatility allows Neumous to effectively respond to the various needs of pharmaceutical companies.
In just three years since its founding, Neumous has gained recognition both domestically and internationally. The company secured a large-scale national project obtaining both funding and technological support, and won the top prize at the K-Startup “King of Kings” Challenge. It was also recognized as an innovative medical device. The Korea Innovative Medicines Consortium (KIMCo) recognizing its achievements, selected Neumous as a company to nurture. Recently, Neumous successfully completed its Series -A funding round. Building on these achievements and promising experimental results, the company now plans to complete clinical trials in a short timeframe and expand onto the global stage.

Dr. Park introduce his technology at the Innovative Medicine Salon in Daejeon / source=Neumous
Neumous is set to complete its clinical trials and release the results globally within this year. Following pre-clinical animal studies, the company confirmed that its technology can open the BBB safely and consistently. Once the clinical results become available, Neumous plans to promptly share its findings with the global pharmaceutical industry and begin the production and supply of devices tailored to client needs.
Dr. Park stated, "Neumous, which began as a research-based initiative, has now grown into a medical device startup with the capability to serve as a partner to pharmaceutical companies. True to our name, we aim to become a company that brings new ‘light’ to patients with brain diseases and advances in brain treatment. We deeply empathize with the suffering of patients and their families, and we are committed to offering them new hope and effective solutions."
By Ju Kyung Cha (racingcar@itdonga.com)







