Excellence of Korean COVID-19 test kits recognized overseas
Excellence of Korean COVID-19 test kits recognized overseas
Posted March. 31, 2020 08:03,
Updated March. 31, 2020 08:03
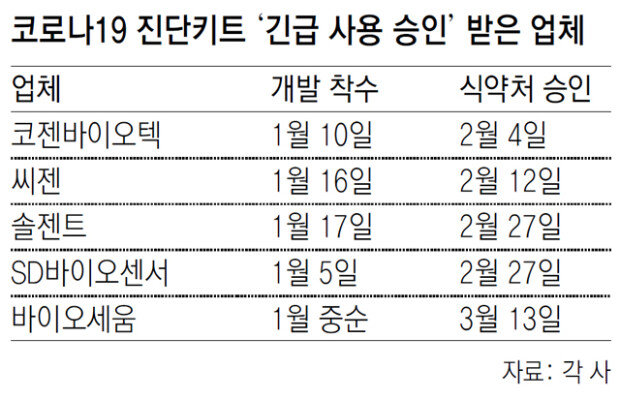
Most of the 117 countries that requested the provision of quarantine items from South Korea for COVID-19 containment are primarily interested in the country’s diagnostic test kits. In fact, experts say that Korean test kits excel both in technology and production capacity, with even the United States asking for assistance from South Korea. How could small-sized developers in South Korea make such strides when the biopharmaceutical giants in advanced economies are still struggling to find the cure?
Experts say that the South Korean companies’ swift decision paid off as they started on the development at an early stage in anticipation of an imminent pandemic. “The major players in Europe were rather sluggish in starting the development, and their slow decision-making was the most critical impediment,” said an official at a local biopharmaceutical company.
Others say that South Korea learned its lessons from the failures of SARS containment. “The spread of the disease brought home the importance of quick testing, and the government supported the development of test kits, prompting the companies begin to grow in earnest,” said a government official. This explains how the local biopharmaceutical developers could advance their technological prowess on the back of a growing number of national research projects and the concomitant expansion of grants.
Hyun-Seok Lim lhs@donga.com







